Clinical trial investigates ‘Light Touch’ approach to wet AMD treatment
News from our full member Moorfields Eye Hospital
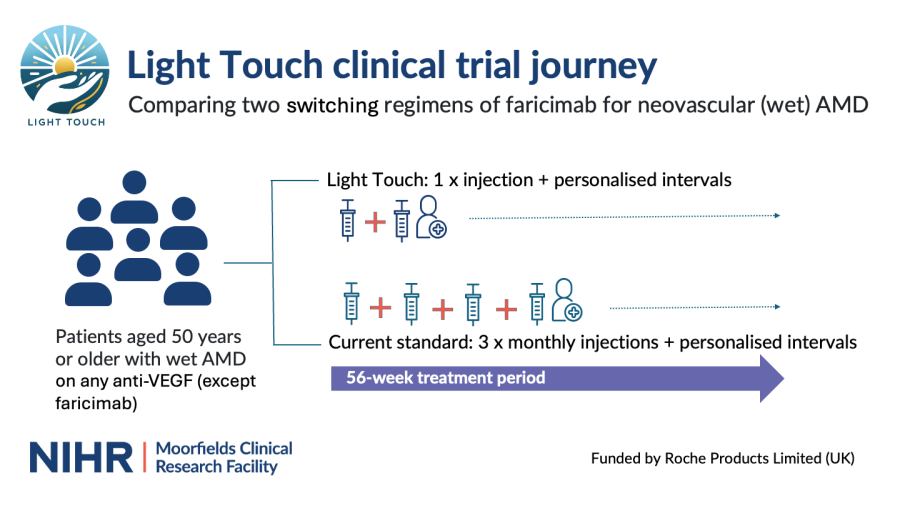
A clinical trial led by NIHR Moorfields Clinical Research Facility will explore a new ‘light-touch’ treatment approach for patients with neovascular age-related macular degeneration (wet AMD) that could result in fewer injections and reduced number of clinic visits.
Funded by Roche, the trial will compare two treatment strategies for patients who have been receiving regular intravitreal injections and are eligible to switch to faricimab (VABYSMO®, Hoffmann-La Roche), an anti-angiogenic agent that patients are increasingly being switched to in order to stem the leaking of retinal blood vessels causing vision loss.
The first strategy, currently used for patients switching over to faricimab, is an initial course of three, monthly intravitreal injections followed by a personalised schedule of injections tailored for the patient according to the level of disease progression.
The second, light touch regimen is a single initial injection at the time of switching over, followed immediately by a personalised schedule of injections.
A phase III clinical trial spanning more than 12 centres, the Light Touch study will assess whether the more streamlined treatment approach is as clinically effective as the traditional three-month induction phase. A positive outcome could lead to a more patient-friendly protocol, maintaining visual outcomes while reducing the burden of frequent injections.
Over the course of two years, 230 patients aged 50 and older with wet AMD will be assessed for the efficacy of the two treatment options. The study will randomize participants into two arms: a standard care group receiving three initial monthly injections and an intervention group receiving a single initial injection with treatment intervals based on their pre-switch treatment schedule.
“This clinical trial represents a significant opportunity to transform how we manage neovascular age-related macular degeneration,” said Dr. Dun Jack Fu, principal investigator at NIHR Moorfields Clinical Research Facility. “By exploring a potentially less intensive treatment approach, we hope to improve patient experience without compromising vision outcomes,” added fellow investigator Dr Livia Faes.
Faricimab, already approved by EMA, MHRA, and NICE for wet AMD treatment, will be used in both study arms. The research will comprehensively evaluate visual acuity improvements, vision loss prevention, anatomical changes, and patient-reported outcomes.
Compared with other licensed agents, which target only one pathway — vascular endothelial growth factor (VEGF) — faricimab takes a dual approach, targeting both the VEGF and angiopoietin-2 pathways, to inhibit leakage of blood vessels in the retina. Farcimab has been found to control disease activity with fewer injections, and longer time intervals between injections. The Light Touch trial seeks to investigate whether even longer time intervals will be clinically effective.
Date: 25 September 2025
Source: Moorfields